van der Heijde D, et al. 2016 update of the ASAS-EULAR management recommendationsfor axial spondyloarthritis.
2016 update of the ASAS-EULAR management recommendationsfor axial SpAを訳します。
会議の後、系統的文献レビュー(SLR)から得られたthe Oxford Centre for Evidence Based Medicineの基準にしたがったエビデンスレベル(LoE)と推奨グレード(GoR)が各の推奨に追加された。まとめるとlevel 1AはRCTsのメタ解析を統合したエビデンスを意味し、Level 1Bは少なくとも一つのRCTに基づくものである。Level 2Aは少なくとも一つの非ランダム化比較試験を意味し、Level 2Bは少なくともひとつの準実験的研究、Level 3は比較対象試験、Correlation studies相関試験やケース・コントロール試験のような記述的研究、Level 4は専門医委員会の報告や尊敬される権力者の意見・経験から来るものを意味する。
The GoRについてはAはLevel 1に一致するという意味、Bはlevel2 or 3の研究に一致するか、level 1の研究の応用、gradeCはlevel 4の研究に基づくか、level 2 or 3の研究からの応用、grade Dはlevel 5 evidenceまたは厄介なほど一貫していないか、いかなるlevelの研究であっても結論がでていないというもの。最終的にtheoverarching principles包括的指針とrecommendations推奨がタスクフォースのメンバーに送られ、各声明に対するlevel of agreement (LoA)を匿名で追加するよう依頼された。LoAはnumerical rating scale (NRS, 0–
Table 1 2016 Update of the ASAS-EULARrecommendations for the management of axSpA

1 axSpA is a potentially severe diseasewith diverse manifestations, usually requiring multidisciplinary managementcoordinated by the rheumatologist.
2 The primary goal of treating the patientwith axSpA is to maximise health-related quality of life through control ofsymptoms and inflammation, prevention of progressive structural damage,preservation/normalisation of function and social participation
3 The optimal management of patients withaxSpA requires a combination of non-pharmacological and pharmacologicaltreatment modalities
4 Treatment of axSpA should aim at the bestcare and must be based on a shared decision between the patient and the rheumatologist
5 axSpA incurs high individual, medical andsocietal costs, all of which should be considered in its management by thetreating rheumatologist
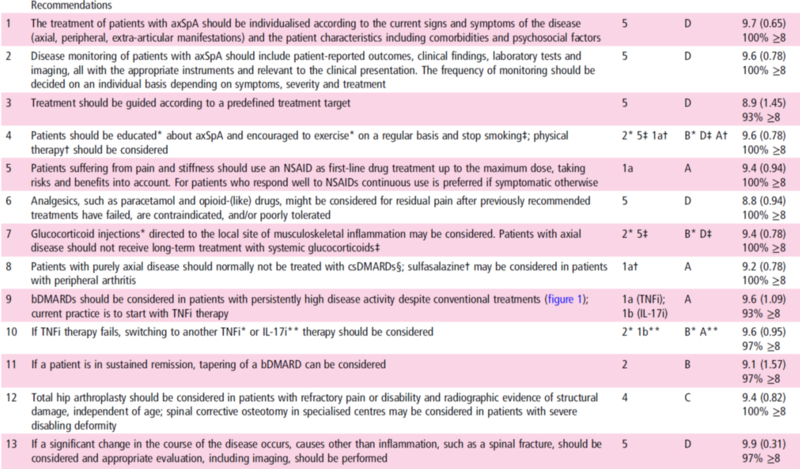
1 The treatment of patients with axSpAshould be individualised according to the current signs and symptoms of thedisease (axial, peripheral, extra-articular manifestations) and the patientcharacteristics including comorbidities and psychosocial factors
2 Disease monitoring of patients with axSpAshould include patient-reported outcomes, clinical findings, laboratory testsand imaging, all with the appropriate instruments and relevant to the clinicalpresentation. The frequency of monitoring should be decided on an individualbasis depending on symptoms, severity and treatment
3 Treatment should be guided according to apredefined treatment target
4 Patients should be educated* about axSpAand encouraged to exercise* on a regular basis and stop smoking‡; physical therapy†should be considered
5 Patients suffering from pain andstiffness should use an NSAID as first-line drug treatment up to the maximumdose, taking risks and benefits into account. For patients who respond well toNSAIDs continuous use is preferred if symptomatic otherwise
6 Analgesics, such as paracetamol andopioid-(like) drugs, might be considered for residual pain after previouslyrecommended treatments have failed, are contraindicated, and/or poorlytoleratedパラセタモールとオピオイド様薬剤のような鎮痛剤は以前に勧められた薬剤で失敗した時、それらが禁忌であった時、あるいは耐用できなかった時に残った疼痛のために考慮されてもよい。
7 Glucocorticoid injections* directed tothe local site of musculoskeletal inflammation may be considered. Patients withaxial disease should not receive long-term treatment with systemicglucocorticoids‡
8 Patients with purely axial disease shouldnormally not be treated with csDMARDs§; sulfasalazine† may be considered inpatients with peripheral arthritis
§1a (sulfasalazine; methotrexate); 1b(leflunomide); 4 other csDMARDs.
9 bDMARDs should be considered in patientswith persistently high disease activity despite conventional treatments (figure1); current practice is to start with TNFi therapy
10 If TNFi therapy fails, switching toanother TNFi* or IL-17i** therapy should be considered
11 If a patient is in sustained remission,tapering of a bDMARD can be considered
12 Total hip arthroplasty should beconsidered in patients with refractory pain or disability and radiographicevidence of structural damage, independent of age; spinal corrective osteotomyin specialised centres may be considered in patients with severe disablingdeformity
13 If a significant change in the course ofthe disease occurs, causes other than inflammation, such as a spinal fracture,should be considered and appropriate evaluation, including imaging, should beperformed
bDMARD, biological disease-modifyingantirheumatic drug;
csDMARD, conventional syntheticdisease-modifying antirheumatic drug;
NSAIDs, non-steroidal anti-inflammatorydrugs;